| Journal of Current Surgery, ISSN 1927-1298 print, 1927-1301 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Curr Surg and Elmer Press Inc |
| Journal website https://www.currentsurgery.org |
Case Report
Volume 12, Number 2, December 2022, pages 45-49
A Large Myofibroblastoma of the Breast in a Premenopausal Woman: A Case Report and Review of the Literature
Zein B. Sheikha, d, Roqaia M. Nafeab, Ahmed S. Alsehlic
aDepartment of Internal Medicine, Prince Mohammed Bin Abdulaziz Hospital, Al Madinah, Saudi Arabia
bDepartment of General Surgery, King Fahd Hospital, Al Madinah, Saudi Arabia
cDepartment of Breast and Endocrine Surgery, King Fahd Hospital, Al Madinah, Saudi Arabia
dCorresponding Author: Zein B. Sheikh, Department of Internal Medicine, Prince Mohammed Bin Abdulaziz Hospital, Al Madinah, Saudi Arabia
Manuscript submitted August 2, 2022, accepted August 29, 2022, published online October 25, 2022
Short title: Large Myofibroblastoma of the Breast
doi: https://doi.org/10.14740/jcs465
| Abstract | ▴Top |
Myofibroblastoma of the breast is a very rare benign mesenchymal tumor, with relatively few cases described in the literature. We report a case of a large mammary myofibroblastoma appearing as a lipoma on ultrasonography. A 47-year-old woman presented with a slowly growing 12-cm mass over a period of 6 years. Imaging studies suggested a diagnosis of lipoma. Tru-Cut biopsy reveled bland spindle cell lesion consistent with myofibroblastoma, which was confirmed after surgical excision. We are reporting this rare entity and reviewed the literature to emphasize myofibroblastoma as a possible differential diagnosis when evaluating a breast mass.
Keywords: Myofibroblastoma; Spindle cell neoplasm; Differential diagnosis
| Introduction | ▴Top |
Myofibroblastoma (MFB) of the breast is a very rare mesenchymal tumor, belonging to the benign spindle cell neoplasms [1, 2]. The incidence of MFB is unknown but estimated to be less than 1% of breast tumors [3]. It was first described as a distinct entity by Wargotz et al in 1987, as a benign tumor of mammary stroma predominantly composed of myofibroblasts [4]. However, Toker et al initially reported four cases of similar tumors in 1981 referring to them as “benign spindle cell tumors of the breast”, possibly being the first to introduce mammary MFB to the literature [5].
MFB primarily occurs in older men and postmenopausal women [1]. Although it has been described earlier with male predominance [4], equal sex distribution has been recently observed [6]. This has been likely attributed to the increase in mammographic screening [7].
Clinically, these tumors present as a slowly growing well-defined mobile breast lump [2]. Mammography and ultrasonography (US) show nonspecific features and can mimic other benign breast lesions [1].
Given the rarity of MFB, along with the nonspecific imaging and cytology features, this tumor can be a cause of diagnostic errors. Final diagnosis requires surgical excision [7].
Here, we report a case of a premenopausal woman with a large MFB of the breast seen as a lipoma on US.
| Case Report | ▴Top |
A 47-year-old Indonesian woman presented to our out-patient clinic complaining of a right breast swelling that was growing slowly over 6 years until reaching a large size, with no history of breast trauma. The patient has regular menstrual cycles, and no previous use of oral contraceptive pills or hormonal therapy. She has no family history of breast cancer. Physical examination revealed a large mass in the upper outer quadrant of the right breast measuring 12 × 10 cm, mobile in respect to the underlying structures but with skin tethering, and no nipple or skin changes. Left breast and both axillae were unremarkable.
Mammography and US showed a circumscribed oval heterogeneous high-density mainly hyperechoic mass in the right breast at 10 o’clock, which had the appearance of a lipoma (Fig. 1). In addition, there were multiple small bilateral benign looking masses for follow-up. No enlarged axillary lymph nodes were noted. It was classified as category 4a in the Breast Imaging Reporting and Data System (BIRADS), therefore we opted to perform the biopsy of the mass.
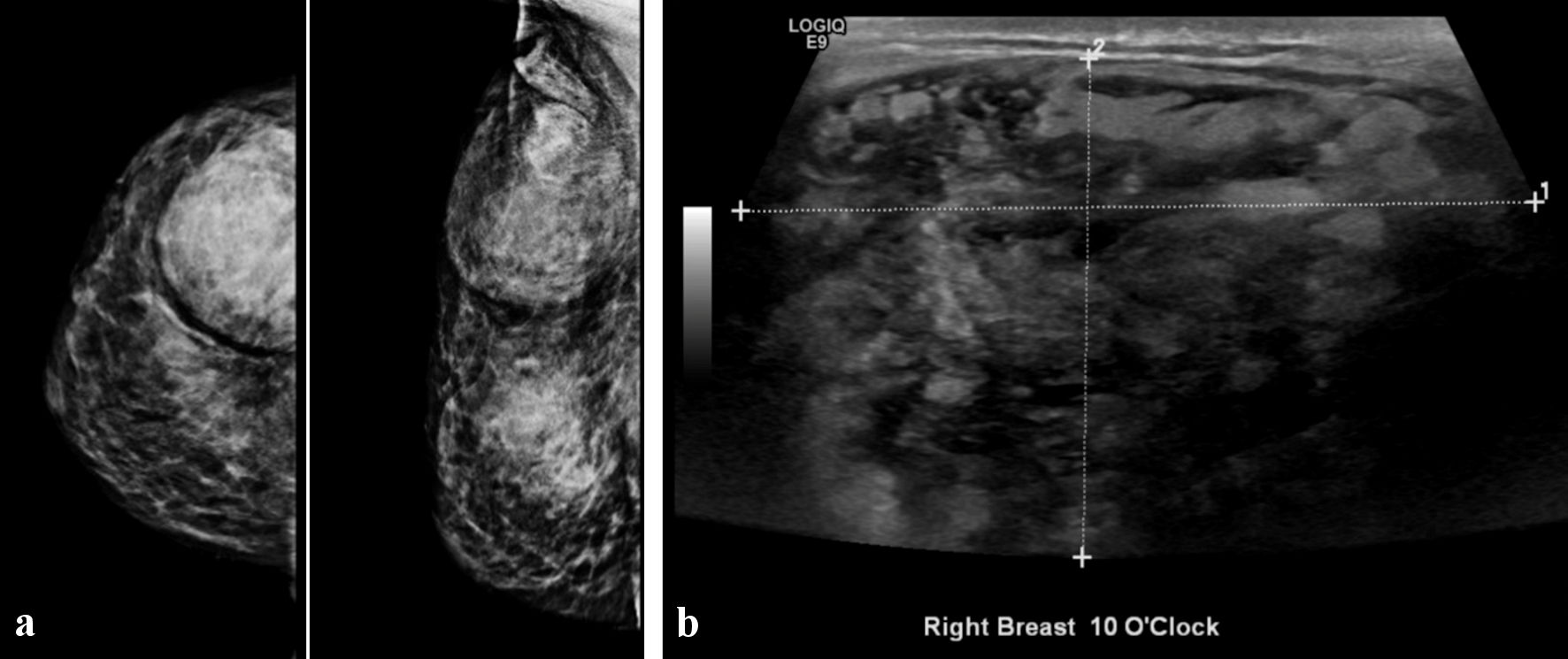 Click for large image | Figure 1. Breast imaging showing circumscribed oval heterogeneous high density mainly hyperechoic mass in the right breast at 10 o’clock. (a) Mammography. (b) Ultrasonography. |
Ultrasound-guided Tru-Cut biopsy showed bland spindle cell lesion consistent with MFB. Immunohistochemistry was positive for estrogen receptor (ER), CD34, desmin, and smooth muscle actin (SMA).
After discussion with the patient and consent taken, we performed surgical excision of the tumor. Resection was done along the capsule with a part of subdermal tissue removed that was adherent to the mass. Patient was discharged home the same day with an uneventful postoperative period. Follow-up duration of 2 years is intended and explained to the patient.
Gross examination revealed a 10 × 8 cm oval shaped mass, well-circumscribed and thinly encapsulated, with a firm-rubbery consistency (Fig. 2). Cut section showed myxoid material mixed with yellowish fatty component, and a grayish-white slimy solid central area (Fig. 3).
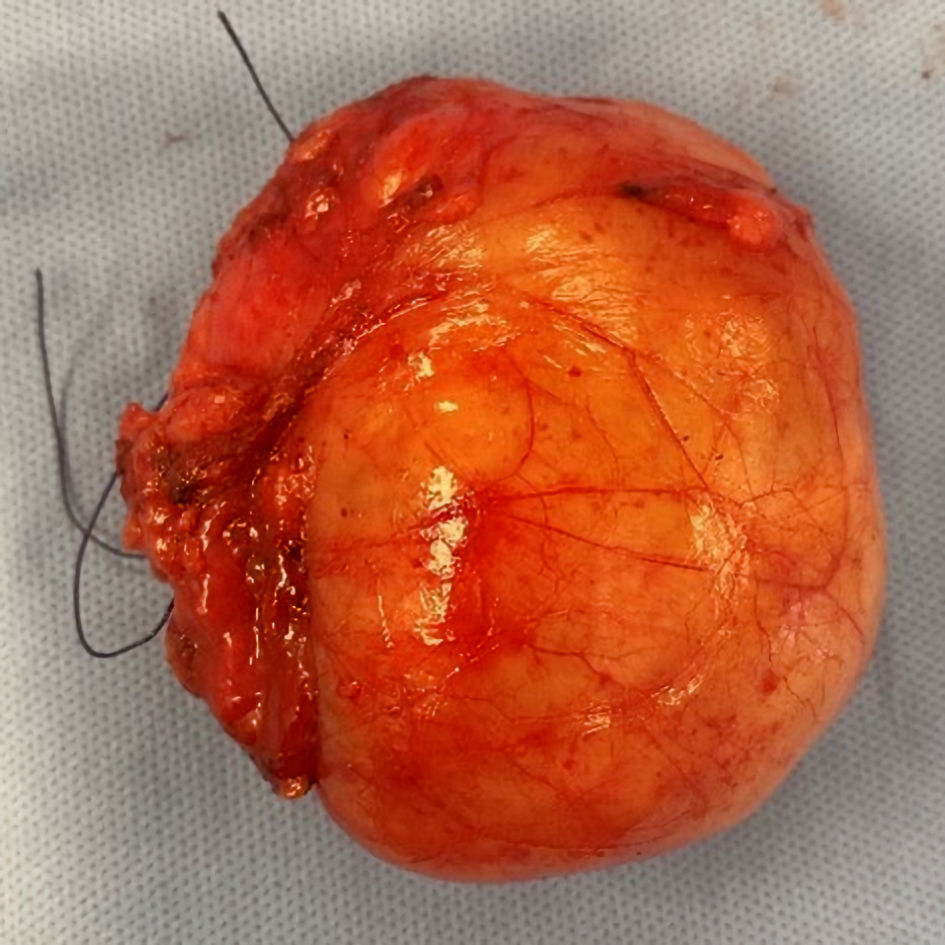 Click for large image | Figure 2. Image of the surgical specimen. |
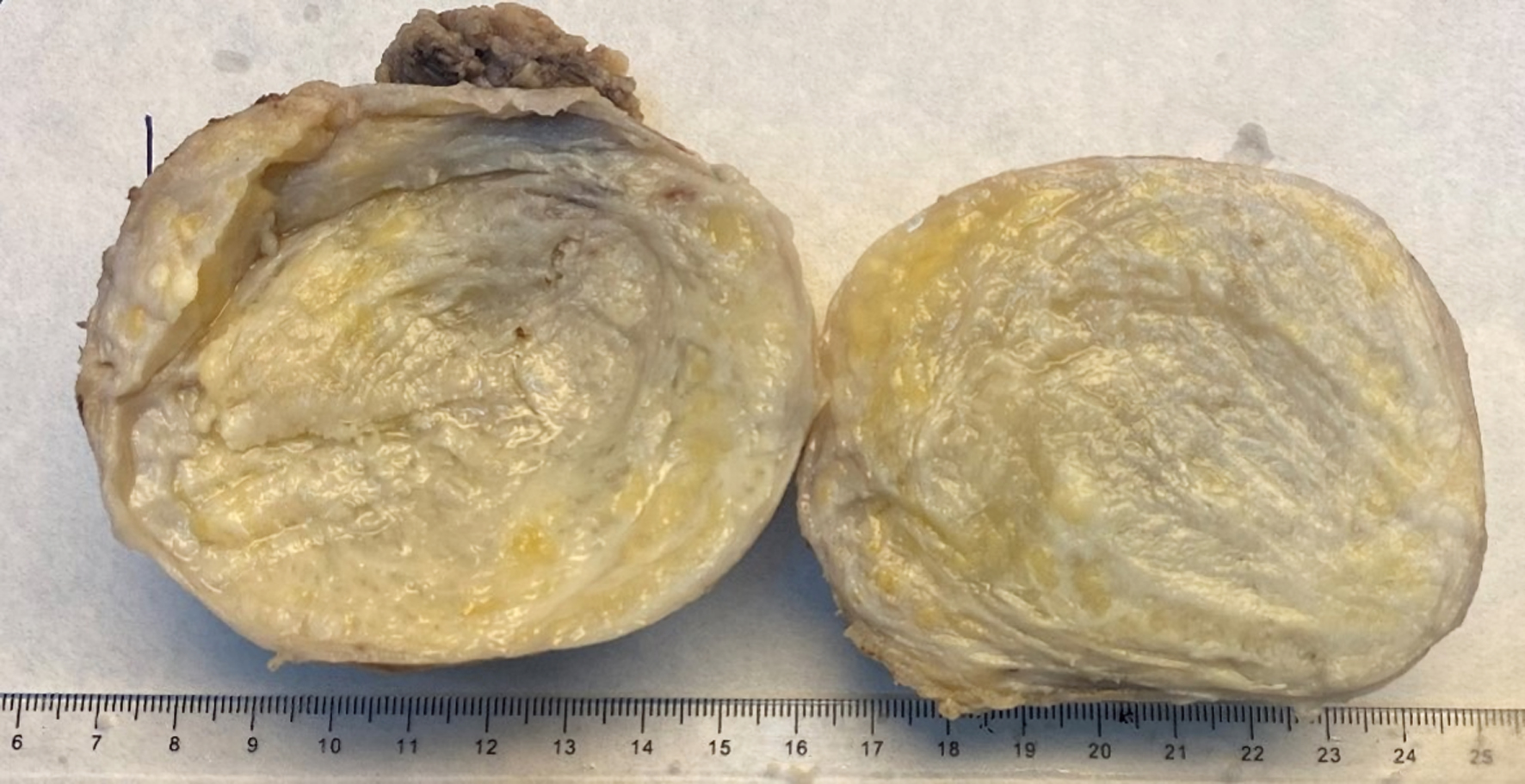 Click for large image | Figure 3. Cut section of the tumor showing myxoid material mixed with yellowish fatty component, and grayish white slimy solid central area. |
Microscopic examination revealed areas of myofibroblastic bipolar spindle cells arranged in haphazard fascicular clusters and stellate cells interspersed with collagenous fibers, adipocytes, and myxoid areas. Scattered mild cellular pleomorphism with hyperchromatic nuclei were noted. No mitotic activity or necrosis were seen (Fig. 4). These findings confirmed the diagnosis of MFB suggested by Tru-Cut biopsy. Small part of subdermal tissue that was adherent to the mass was sent for microscopic evaluation, which showed no tumor cell invasion.
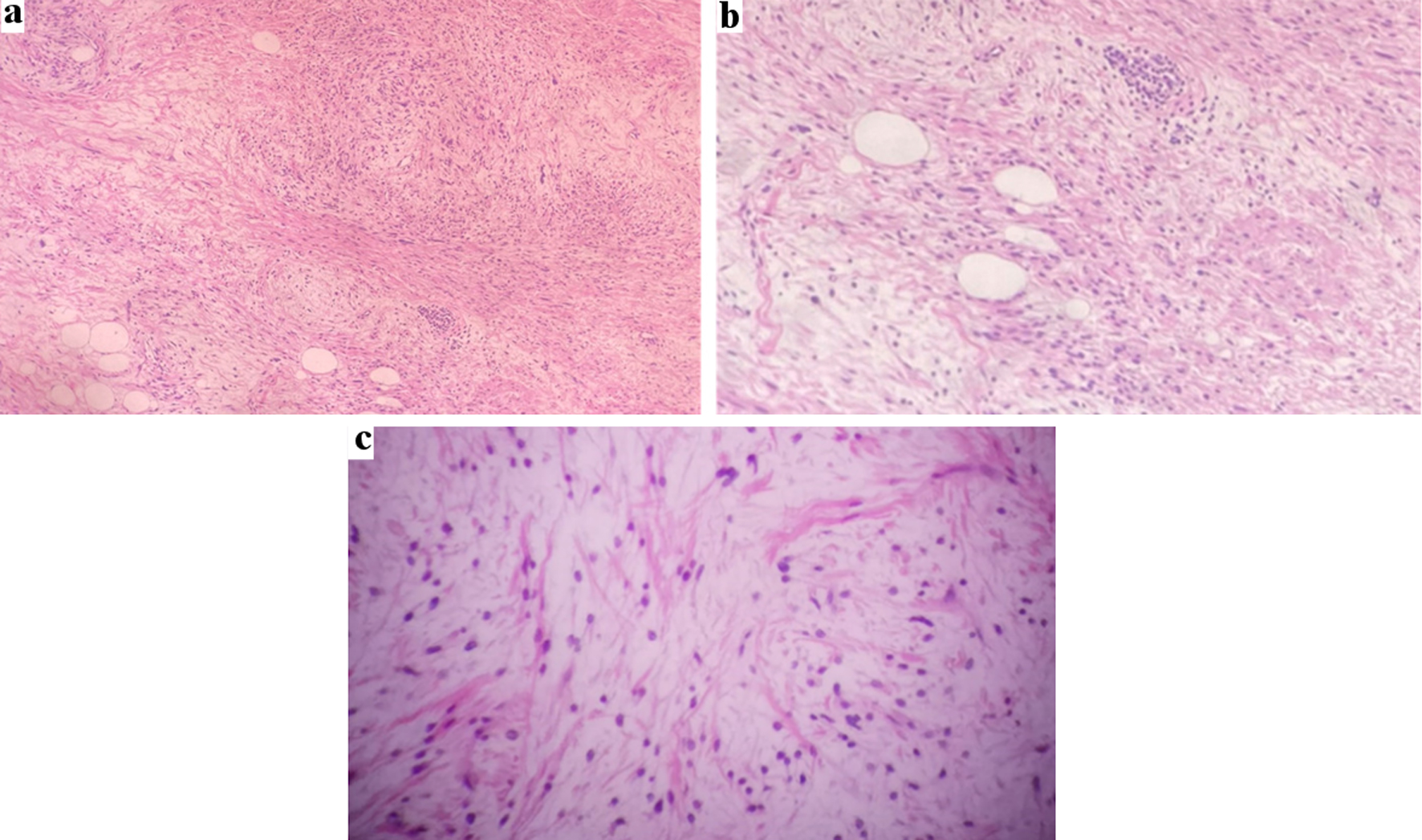 Click for large image | Figure 4. (a) Microscopic examination showing myofibroblastic spindle cell clusters and stellate cells interspersed with collagenous fibers, adipocytes, and myxoid areas. (b) Higher magnification scattered mild cellular pleomorphism with hyperchromatic nuclei. (c) Higher magnification, bipolar spindle cells arranged in intersecting fascicles in focal myxoid area. |
| Discussion | ▴Top |
MFB is a very uncommon benign tumor. Although it can arise from various sites including skin, soft tissue, and lymph nodes, mammary MFB is the most common [3, 7]. It is a mesenchymal tumor that originates from the breast stroma and belongs to the mammary benign spindle cell neoplasms [1]. These are a group of tumors that consist of spindle-shaped cells histologically [8], and include benign fibroblastic spindle cell tumor, leiomyoma, schwannoma, spindle cell lipoma, solitary fibrous tumor, and myxoma [9].
Clinical presentation usually mimics fibroadenoma [10] with a solitary painless well-defined mobile mass, which grows slowly over months to several years [2]. Very few cases have reported adherence to the skin [1, 3, 11]. In our case, the tumor was tethered to the skin clinically, but was able to be separated from the subdermal tissue in the operating room, and histopathologic evaluation confirmed no invasion to subdermal tissue. Recently, cases with non-palpable masses discovered through mammographic screening have been described [7].
Breast imaging cannot differentiate MFB from other benign breast lesions like fibroadenoma, hamartoma, or in some instances similar to this reported case, lipoma [1]. Mammography typically shows a well-circumscribed dense mass without calcifications, and US demonstrates a well-circumscribed often mainly hypoechoic mass [12]. However, variable degrees of acoustic attenuation can be seen according to the type of tissue incorporated in the tumor [1]. In our case, US displayed a mainly hyperechogenic mass and was initially described as a possible lipoma. Therefore, the nonspecific imaging features of MFB warrant tissue sampling for diagnosis [10].
Although fine needle aspiration cytology (FNAC) can suspect MFB, misdiagnosis with phyllodes tumor or malignancy can occur [12]. Tru-Cut biopsy has been found to be a reliable method for preoperative diagnosis [1]. Nonetheless, pathologists can face diagnostic difficulty in some cases, especially those with unusual morphologic variants [12].
Grossly, MFB is well-circumscribed, unencapsulated, firm-rubbery in consistency, with a rounded to oval shape [12]. In the present reported case, the tumor was encapsulated with a thin well-delineated capsule that was easily separated from the mass. The average size of these tumors ranges between 1 and 3.7 cm [13], and large MFBs of 10 cm and above have been rarely reported [14]. The tumor in our case has grown to a 10-cm mass. Cut section shows solid tumor that is white to gray in color, with smooth or lobulated surface, and in some cases show focal areas of lipomatous or extensive mucoid appearance [10].
Microscopically, classic type MFB appears as bland bipolar spindle cells arranged in haphazard short fascicles interrupted by dense hyalinized collagen bundles, with low mitotic count, and no atypical mitosis or necrosis [15]. Myxoid changes or lipomatous component may be variably seen in a minority of cases [12], which is found in the present report. Recently, a wide spectrum of histologic variants has been recognized, which include cellular, epithelioid, deciduoid-like, collagenized/fibrous, infiltrative, lipomatous, myxoid, and mixed variants (Table 1) [10]. Although the tumor in our case displayed a lipomatous component, only cases that are composed predominantly (i.e., > 75% of the entire neoplasm) of adipocytes are classified as lipomatous MFB. Similarly, while our tumor had areas of myxoid changes, the term myxoid MFB is reserved for tumors consisting entirely or predominantly of myxoid stroma [12].
 Click to view | Table 1. Morphologic Variants of Myofibroblastoma |
Light microscopy alone can easily diagnose MFB in the surgical specimen; however, immunohistochemistry is very important in excluding other benign and malignant spindle cell neoplasms. Typically, MFB displays positivity for CD34, desmin, vimentin, estrogen, and progesterone. Other cases show frequent but variable positivity for SMA, actin, bcl-2, and CD99. Meanwhile, MFB is consistently negative for cytokeratins, S100 protein, EMA, HMB-45, and c-Kit (CD117) [12]. In this report, the tumor was positive for CD34, desmin, SMA, and ER, most consistent with MFB. Immunoreactivity for desmin excluded spindle cell lipoma as a differential diagnosis, while CD34 positivity excludes fibromatosis.
Surgical excision with free resection margin is curative [1], and recurrence is unlikely even with positive margins [16]. No malignant transformation has been reported to date. Nonetheless, a minimum follow-up period of 2 years is advised [1].
Conclusions
MFB of the breast is a very rare benign mesenchymal tumor. It usually affects older men and postmenopausal women but can present in a younger age. Imaging studies cannot differentiate MFB from other benign lesions, and FNA can lead to diagnostic error. Core needle biopsy is a reliable method for diagnosis and should be interpreted carefully to avoid misdiagnosis with malignancy. Local excision is curable with no reported cases of malignant transformation, and very few recurrence cases reported in the literature. Minimum follow-up period of 2 years is recommended. Clinicians should be aware of this diagnosis when interpreting radiology and biopsy results in a patient presenting with a breast mass to avoid diagnostic errors.
Acknowledgments
The authors would like to acknowledge the input of Dr. Syed N Ahmed in the histopathology section of this case report.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Consent has been taken.
Author Contributions
ZS performed data collection and manuscript writing. RN performed case management, critical revision of the manuscript, and final approval. AA performed case management, decision making, critical revision of the manuscript, and final approval.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
MFB: myofibroblastoma; US: ultrasonography; BIRADS: Breast Imaging Reporting and Data System; ER: estrogen receptor; SMA: smooth muscle actin; FNAC: fine needle aspiration cytology
| References | ▴Top |
- Mele M, Jensen V, Wronecki A, Lelkaitis G. Myofibroblastoma of the breast: case report and literature review. Int J Surg Case Rep. 2011;2(6):93-96.
doi pubmed - Metry M, Shaaban M, Youssef M, Carr M. Myofibroblastoma of the breast: literature review and case report. Case Rep Oncol Med. 2016;2016:1714382.
doi pubmed - Abdalla O, Chitnis A, Omer B, Elhadi M, Karim MO, Sabri S. A case report of successful treatment of mammary myofibromyoblastoma in a male patient. Journal of Current Surgery. 2019;9(2-3):26-28.
doi - Wargotz ES, Weiss SW, Norris HJ. Myofibroblastoma of the breast. Sixteen cases of a distinctive benign mesenchymal tumor. Am J Surg Pathol. 1987;11(7):493-502.
doi pubmed - Toker C, Tang CK, Whitely JF, Berkheiser SW, Rachman R. Benign spindle cell breast tumor. Cancer. 1981;48(7):1615-1622.
doi - Bharathi K, Chandrasekar VA, Hemanathan G, Anuradha S. Myofibroblastoma of female breast masquerading as schirrous malignancy - a rare case report with review of literature. J Clin Diagn Res. 2014;8(6):ND10-11.
doi pubmed - Khatib Y, Pandey V, Khade AL, Pandey R. Myofibroblastoma of the breast: a rare cause of breast lump in a postmenopausal woman. J Midlife Health. 2018;9(1):47-49.
doi pubmed - Shamim T. The spindle cell neoplasms of the oral cavity. Iran J Pathol. 2015;10(3):175-184.
- Magro G. Differential diagnosis of benign spindle cell lesions. Surg Pathol Clin. 2018;11(1):91-121.
doi pubmed - Talu CK, Cakir Y, Hacihasanoglu E, Leblebici C, Aksoy S, Nazli MA. Inflammatory myofibroblastic tumor of the breast coexisting with pseudoangiomatous stromal hyperplasia. J Breast Health. 2016;12(4):171-173.
doi pubmed - Mnif H, Charfi S, Abid N, Sallemi-Boudawara T. Mammary myofibroblastoma with leiomyomatous differentiation: case report and literature review. Pathologica. 2013;105(4):142-145.
- Magro G. Mammary myofibroblastoma: a tumor with a wide morphologic spectrum. Arch Pathol Lab Med. 2008;132(11):1813-1820.
doi pubmed - Zahid MF, Zafar I, Din NU, Ahmed A, Fatima S, Kayani N. Mammary myofibroblastoma: a clinico- pathologic study of six cases. Breast Dis. 2015;35(2):143-148.
doi pubmed - Yan M, Bomeisl P, Gilmore H, Sieck L, Kuchta Z, Harbhajanka A. Clinicopathological and radiological characterization of myofibroblastoma of breast: A single institutional case review. Ann Diagn Pathol. 2020;48:151591.
doi pubmed - Magro G. Mammary myofibroblastoma: an update with emphasis on the most diagnostically challenging variants. Histol Histopathol. 2016;31(1):1-23.
- Howitt BE, Fletcher CD. Mammary-type myofibroblastoma: clinicopathologic characterization in a series of 143 cases. Am J Surg Pathol. 2016;40(3):361-367.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Current Surgery is published by Elmer Press Inc.
